As all of the other substances are soluble in water we can rewrite the equation.\) formula unit on the right side.īy eliminating the spectator ions, we can focus on the chemistry that takes place in a solution. 
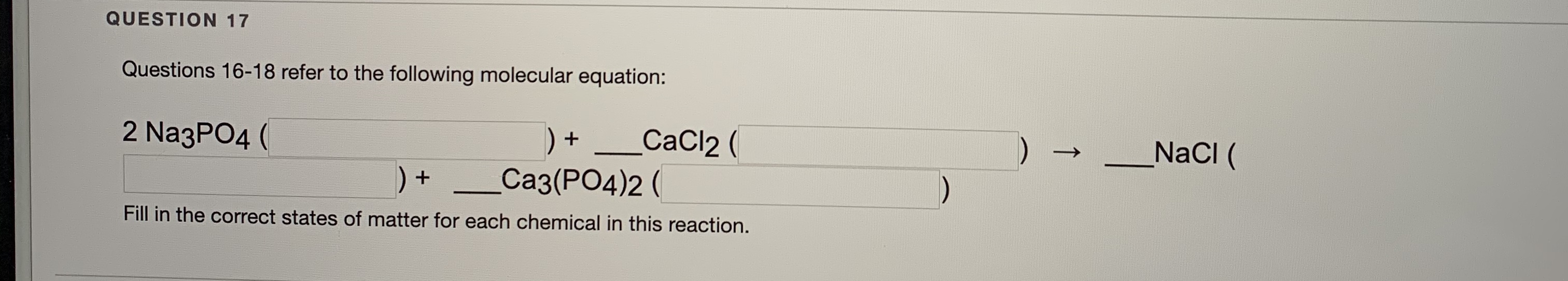
This is an example of a double displacement reaction. Precipitation and double displacement reaction. Because it is insoluble in water we know that it is the precipitate. Cobalt phosphate precipitate (Co3(PO4)2) is formed as a result: CoCl2 + Na3PO4 -> Co3(PO4)2 + NaCl. This video is the practical demonstration of the reaction of Sodium phosphate (Na3PO4) with Calcium chloride (CaCl2). We would expect them to undergo a double displacement reaction with each other.īy examining the solubility rules we see that, while most sulfates are soluble, barium sulfate is not. Approximately 2 mL of Solution A (on the left) is added to a sample of Solution B (on the right) with a dropping pipet. The initial blue color of the combined solution fades. Write the reaction and identify the precipitate.īarium chloride and potassium sulfate are both ionic compounds. A blue-green solution of copper(II) chloride is combined with a colorless solution of sodium phosphate. Pricipitate forms Precipitate does not form NaNO3 + NaBr Fe (NO3)2 + Na2SO4 AgNO3 + NaC104 AgNO3 + NaCl Mg (NO3)2 + NaOH Pb (NO3)2 + Nal Fe (NO3)2 + Na3PO4 Answer Bank. The exceptions are the alkali metals and the ammonium ion.ĬaSO 4 and Ag 2SO 4 are slightly soluble.Ī solution of barium chloride is mixed with a solution of potassium sulfate and a precipitate forms. A student wants to determine if PO43 is part of. Carbonates (CO 3 -2), phosphates (PO 4 -3) and sulfides (S -2) are insoluble. Mg3(PO4)2 MgPO4 Na Mg O NaCl NaCl2 QUESTION 8 Solutions of ZnCl2 and Na3PO4 are mixed and a precipitate forms. When a colorless solution of silver nitrate is mixed with a yellow-orange solution of potassium dichromate a reddish precipitate of silver dichromate is produced. 5 drops of 1 M nitric acid, HNO3, to about 10 drops of 0.5 M sodium chloride, NaCl. A precipitation reaction is a reaction that yields an insoluble producta precipitatewhen two solutions are mixed. The exceptions are those containing Ag +, Hg +2, and Pb +2.Ħ. The solution is acidic when the blue litmus changes to a red color). Most chlorides (Cl -), bromides (Br -) or iodides (I -) are soluble. The exceptions are the alkali metal hydroxides and Ba(OH) 2.ĥ. Nitrates (NO 3 -), chlorates (ClO 3 -), and perchlorates (ClO 4 -) are soluble. Balanced Chemical Reaction Equation with reactants na3po4 (sodium phosphate) cacl2 (calcium chloride) and products ca3(po4)2 (calcium phosphate) nacl (sodium chloride). Ammonium (NH 4 +) compounds are soluble.ģ. Will a precipitate form when 100.0 mL of a 0.200 M Fe(NO3)2 solution is mixed with 25.0 mL of a 0.400 M NaOH solution Explain. Alkali metal (Group IA) compounds are soluble.Ģ. Solubility Rules and Identifying a Precipitateġ. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
